3. The systems acetone–sodium hydroxide–water and acetone–potassium hydroxide–water at 0° - Journal of the Chemical Society (Resumed) (RSC Publishing)

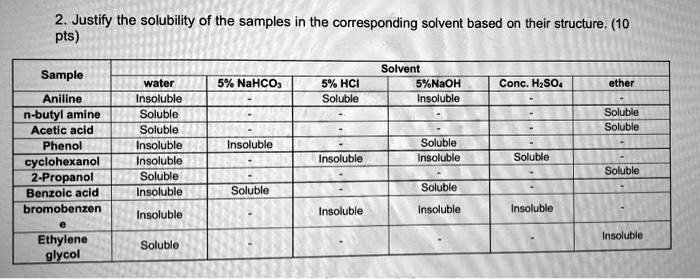
SOLVED: Four test tubes containing different hydrocarbons: acetone, benzoic acid, cyclohexane, and ethylamine were tested for solubility with different solvents: H2O, dilute HCl solution, and a strong solution of NaOH. The results
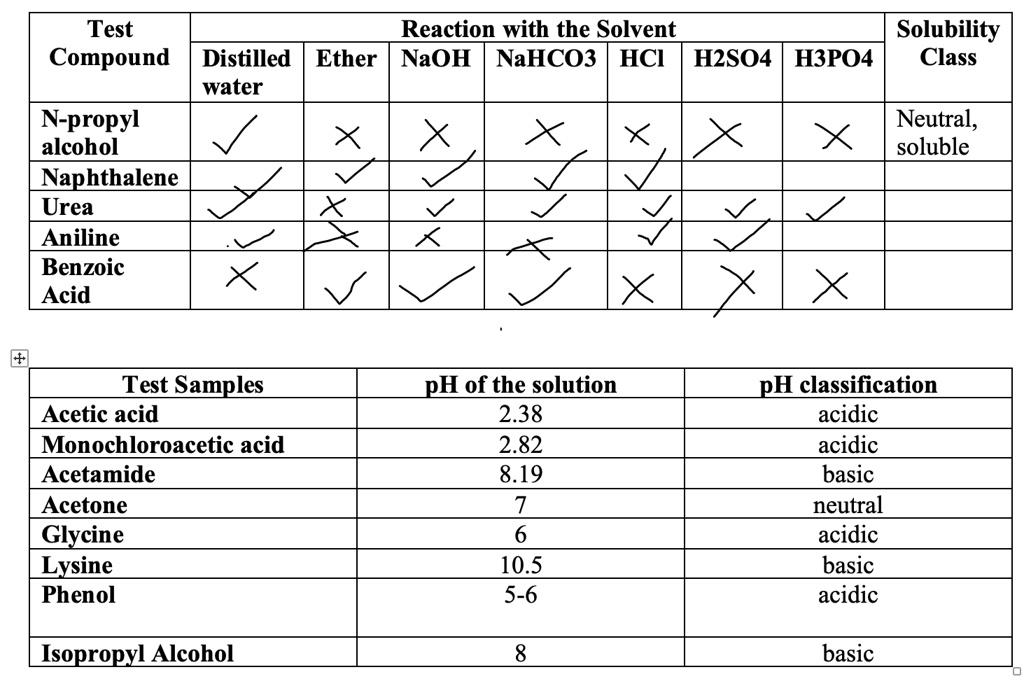
SOLVED: Test Reaction with the Solvent Solubility Compound Distilled Ether NaOH NaHCO3 HCI H2SO4 H3PO4 Class water N-propyl Neutral alcohol soluble Naphthalene Urea Aniline Benzoic Acid Test Samples Acetic acid Monochloroacetic acid

What reactions result from the addition of sodium hydroxide to the mixture of acetone and vanillin? | Homework.Study.com

Effects of NaOH and Ethanol on the Solubility of Disodium Decanedioate in Water | Journal of Chemical & Engineering Data

Solubility of Electrolytes in Organic Solvents: Solvent-Specific Effects and Ion-Specific Effects | Journal of Chemical & Engineering Data
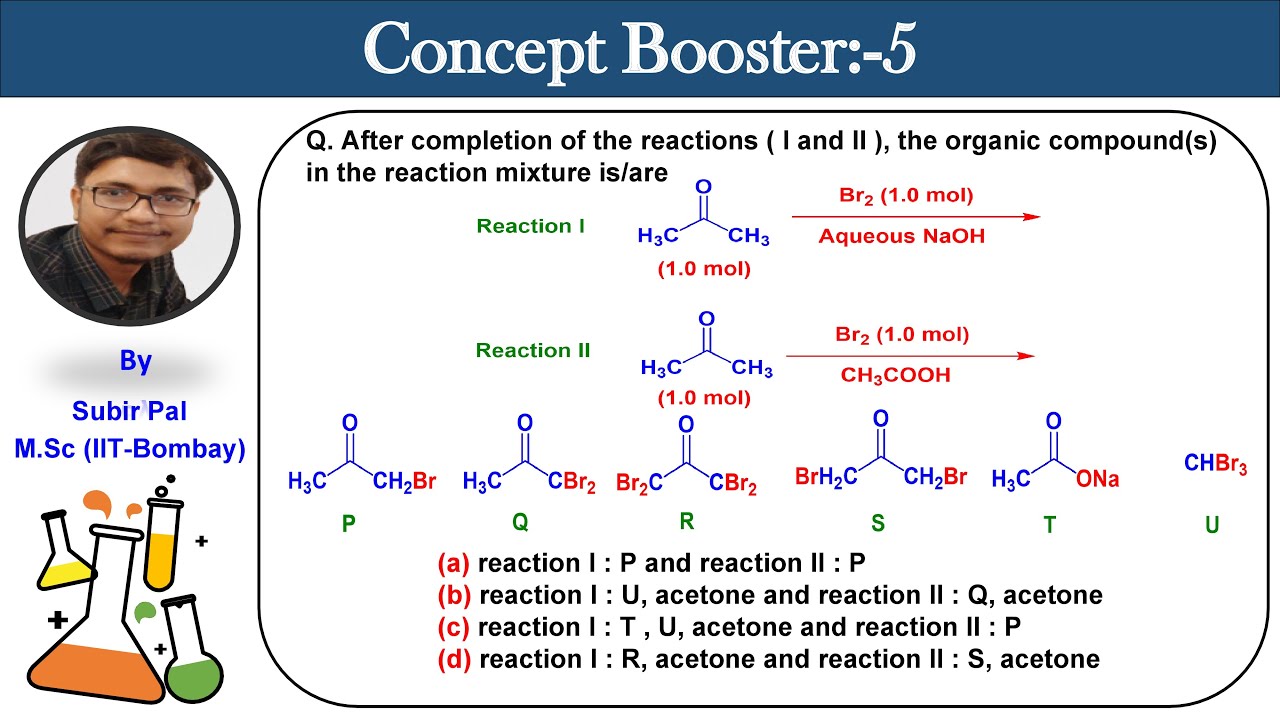
Bromination of Acetone in Aqueous NaOH and CH3COOH || Concept Booster-5 || CSIR-NET || GATE - YouTube