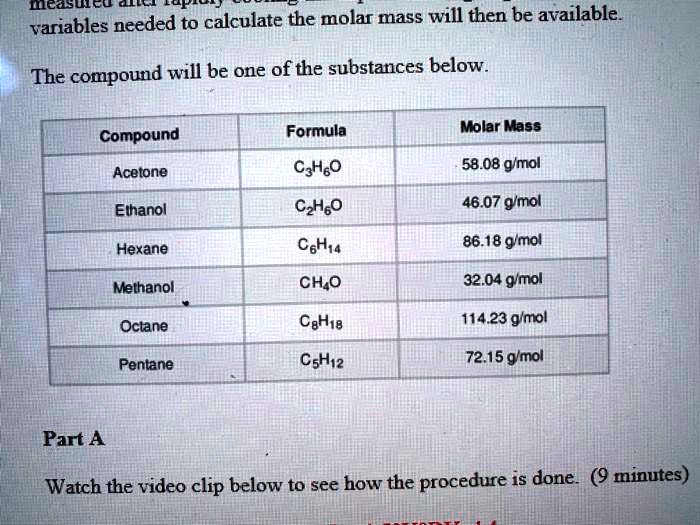
SOLVED: Molecules 4 variables needed to calculate the molar mass will then be available: The compound will be one of the substances below Formula Molar Mass Compound Acetone C3H6O 58.08 g/mol Ethanol

SOLVED: How much energy is required to heat 87.1 g of acetone (molar mass = 58.08 g/mol) from a solid at -154.0°C to a liquid at -42.0°C? The following physical data may

A student determines the molar mass of acetone, CH,COCH, by the method used thum found that the equilibrium temperature of a mixture of ice and water was 1.0 mixture of ice and

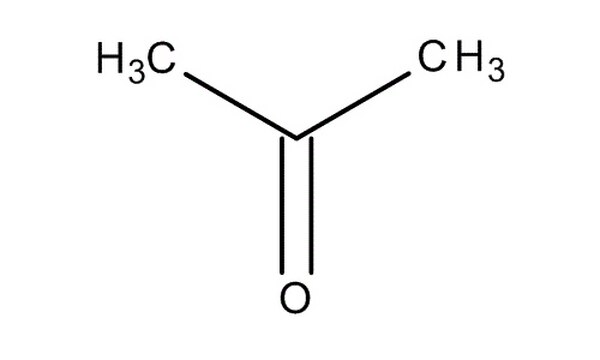
Acetone, or propanone, is an organic compound with the formula (CH3)2CO. It is the simplest and smallest ketone. It is … | Chemical structure, Chemistry, Molar mass

SOLVED: Calculate the mass of acetone, C3H6O, that vaporizes as a result of absorbing 500 kJ of energy. The molar enthalpy of vaporization of acetone is 31.3 kJ/mol.

SOLVED: A chemist wants to determine the molar mass of a naturally occurring substance isolated from spearmint. She adds 4.531 g of this substance to 25.00 g of acetone (C3H6O; molar mass
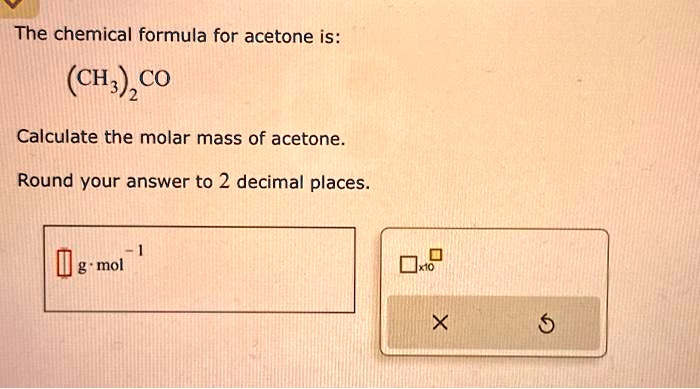
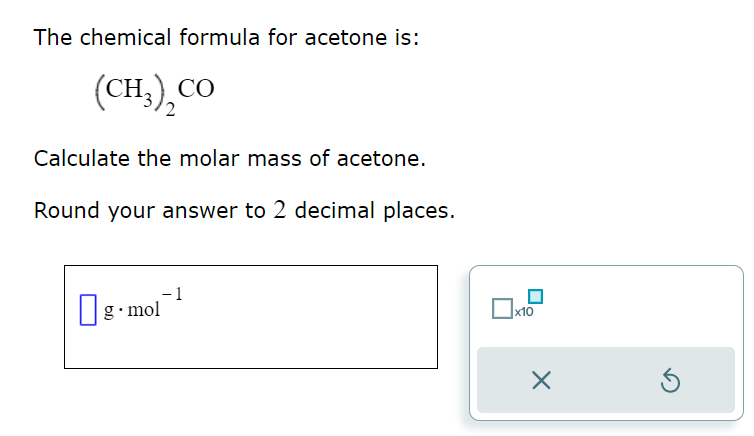
SOLVED: The chemical formula for acetone is: (CH),CO Calculate the molar mass of acetone Round your answer to 2 decimal places. g.mol
✓ Solved: Calculate the molarity and mole fraction of acetone in a 1.00-m solution of acetone (CH3 COCH3)...

SOLVED: What is the mass in grams of 0.935 mol of acetone; CH;COCH3? The molar mass of acetone is 58.08 g/mol. A. 0.0161 g B. 54.3 g C. 57.1 g D. 62.1 g






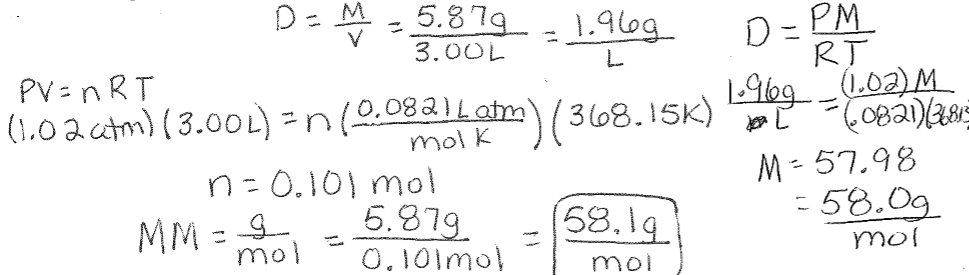
![Tamil] Calculate the molar mass of the following compounds. (ii) Ac Tamil] Calculate the molar mass of the following compounds. (ii) Ac](https://static.doubtnut.com/ss/web-overlay-thumb/3190387.webp)

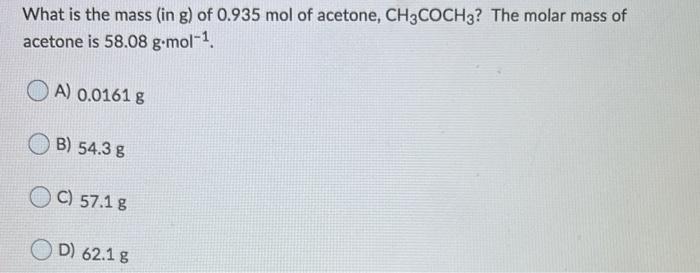
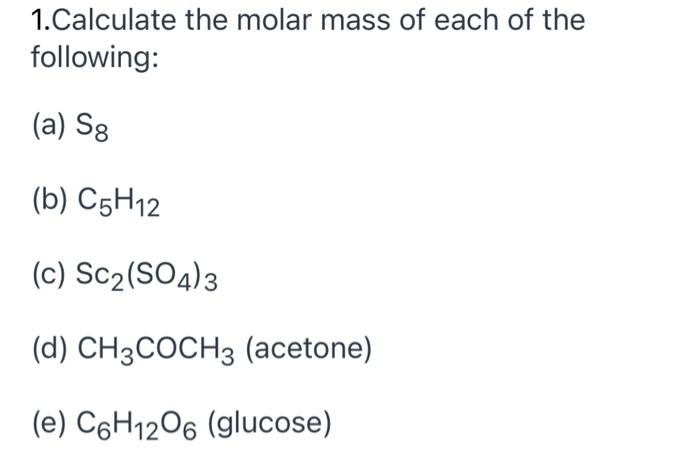
![Acetone [(CH3)2CO] Molecular Weight Calculation - Laboratory Notes Acetone [(CH3)2CO] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2023/02/acetone-molecular-weight-calculation-300x200.jpg)
