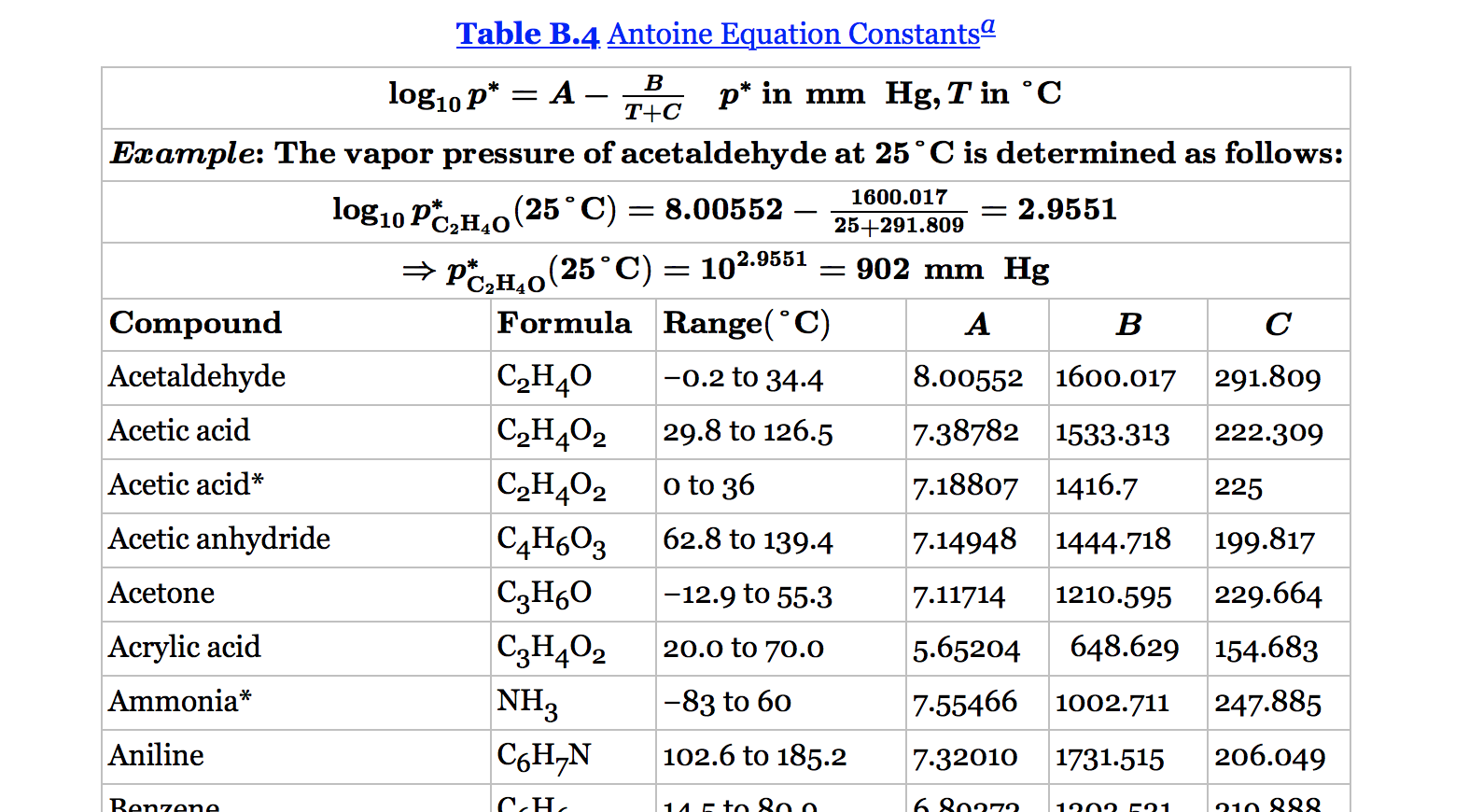
Vapor pressures (bar) calculated by the extended Antoine equation and... | Download Scientific Diagram

SOLVED: The vapor pressure of acetone (labeled as acetonitrile 2) and nitromethane 3 can be represented by the Antoine equation as: 2795.82 ln(Psat) = 14.3916T + 230 2945.47 ln(Psat) = 14.2724T +

APPENDIX B.2. Constants For The Antoine Equation For Vapor Pressures of Pure Species | PDF | Ethanol | Chemistry

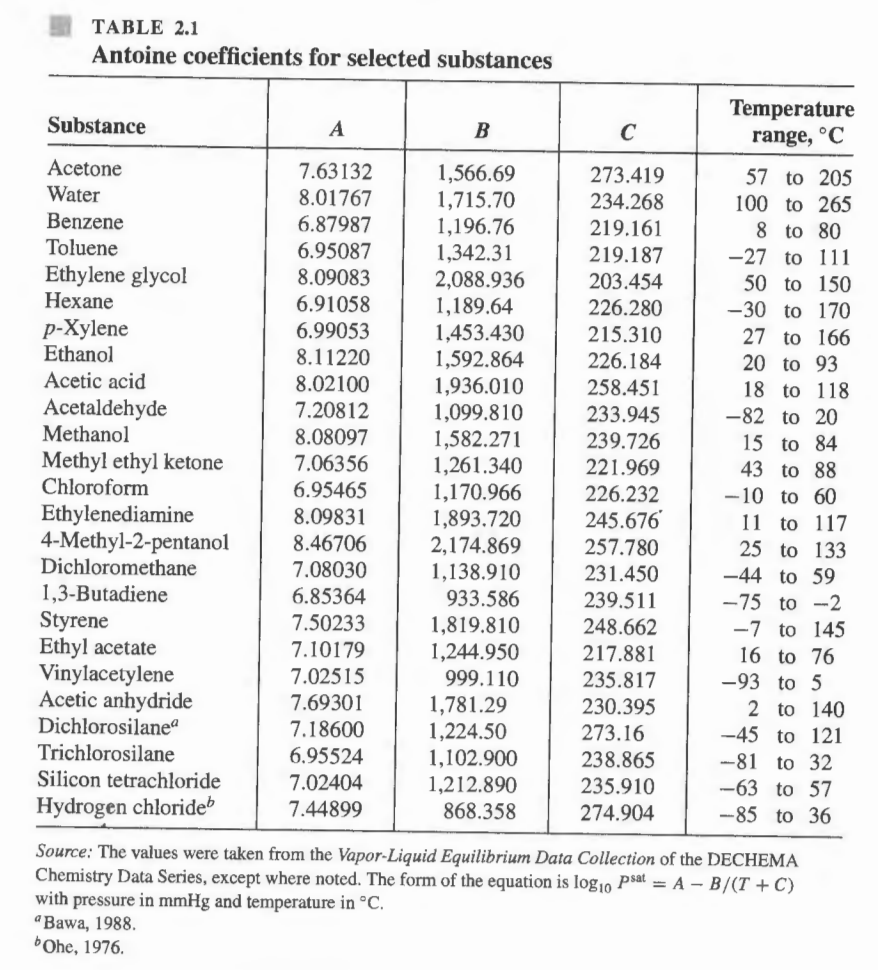



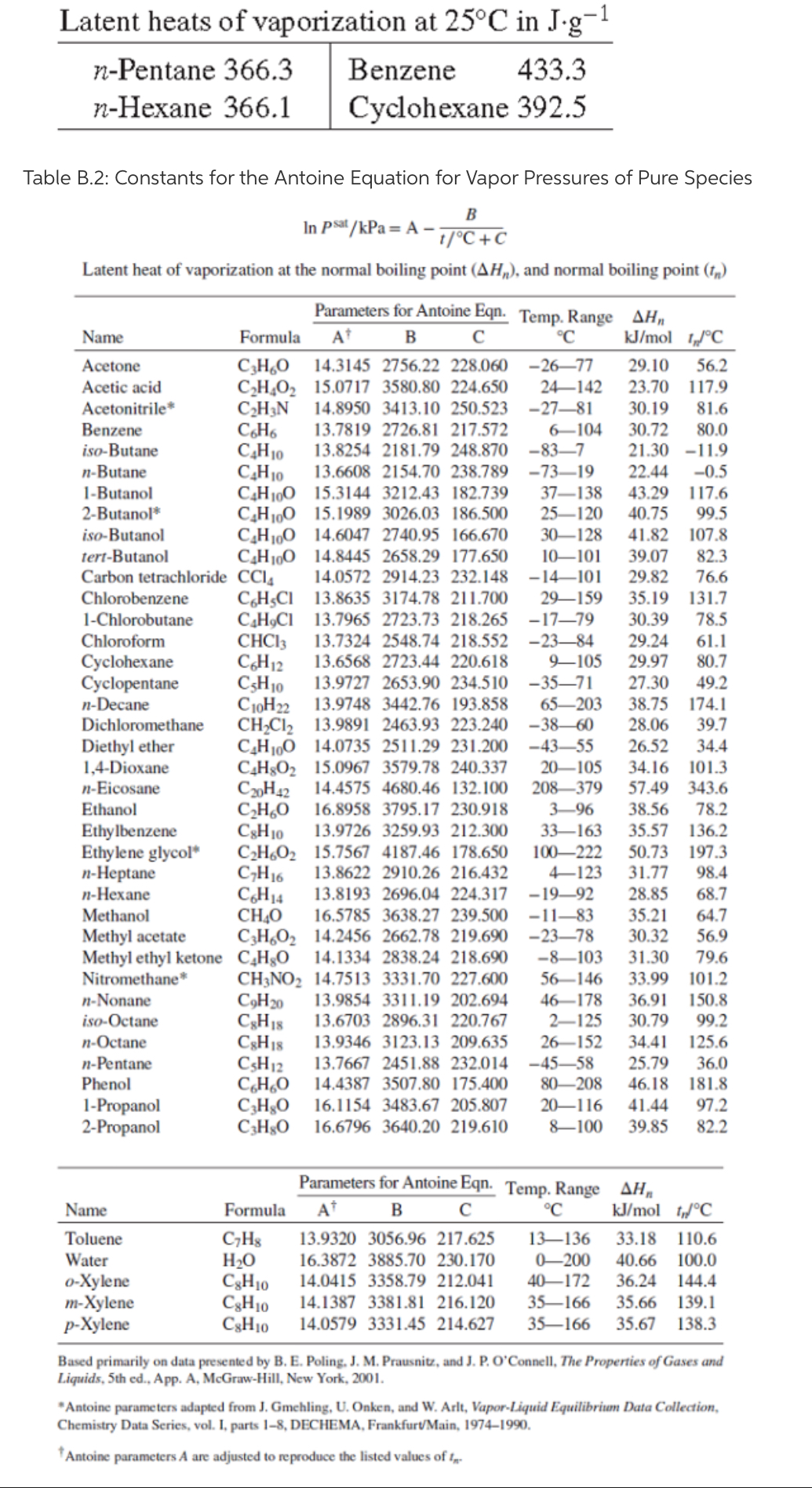






![Solved] For the acetone(1)/methanol(2)/water(3) s | SolutionInn Solved] For the acetone(1)/methanol(2)/water(3) s | SolutionInn](https://dsd5zvtm8ll6.cloudfront.net/si.question.images/images/question_images/1646/8/3/0/0306228a1ce1f7e31646830024159.jpg)