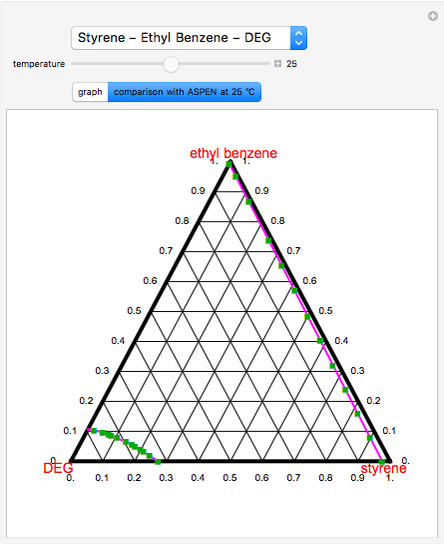
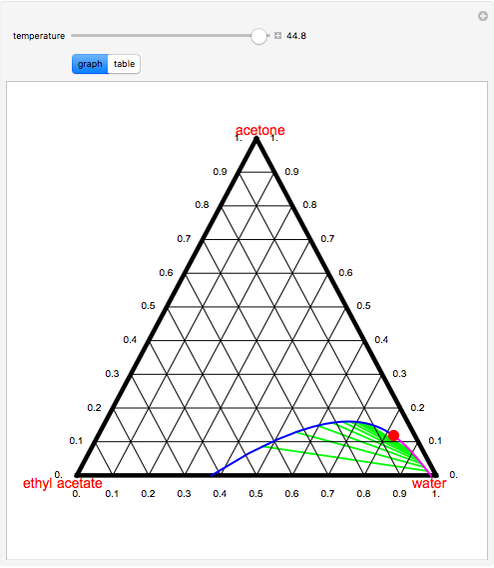
Polymers | Free Full-Text | Effect of Acetone as Co-Solvent on Fabrication of Polyacrylonitrile Ultrafiltration Membranes by Non-Solvent Induced Phase Separation
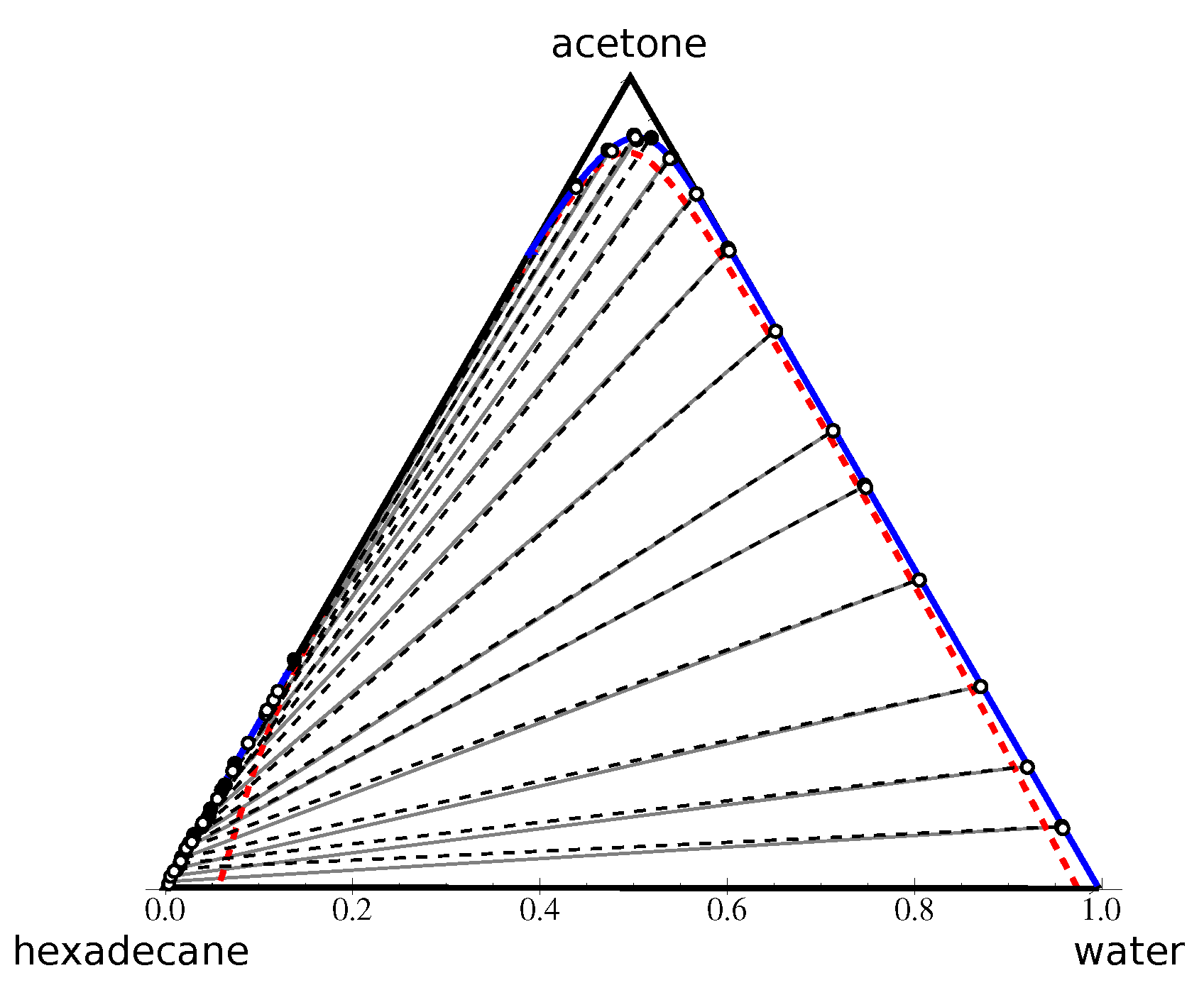
Entropy | Free Full-Text | Using the Intrinsic Geometry of Binodal Curves to Simplify the Computation of Ternary Liquid–Liquid Phase Diagrams

Physical properties and their corresponding changes of mixing for the ternary mixture acetone + n-hexane + water at 298.15 K - ScienceDirect
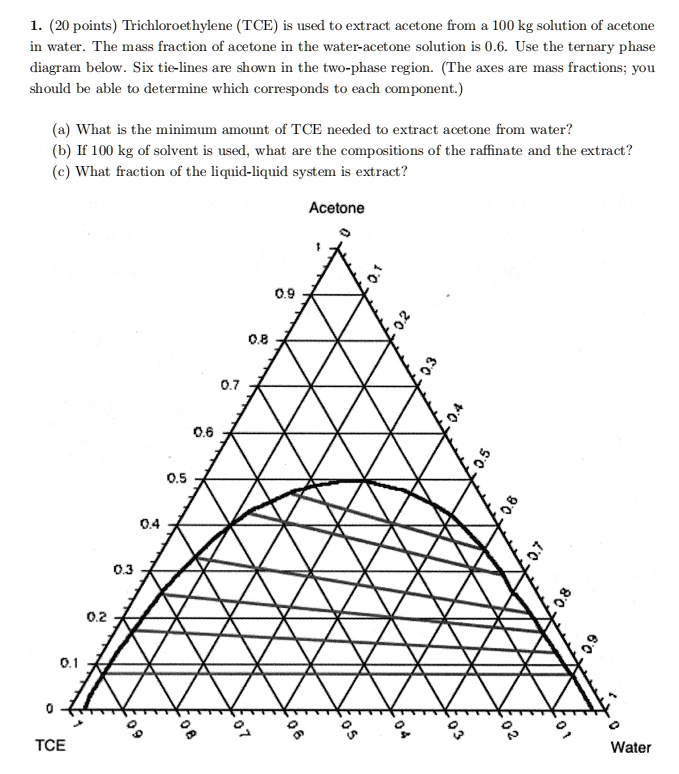
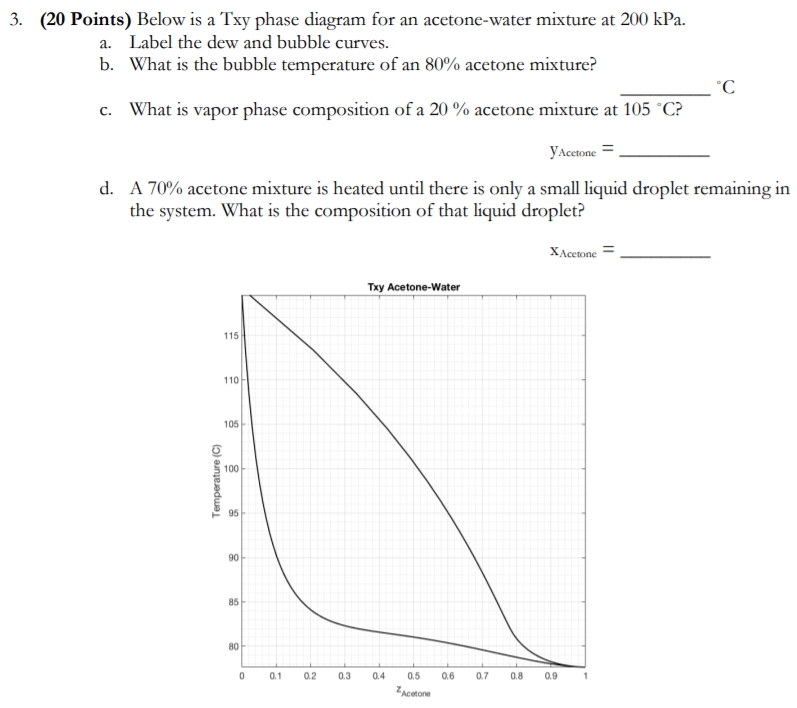
SOLVED: Trichloroethylene (TCE) is used to extract acetone from a 100 kg solution of acetone in water. The mass fraction of acetone in the water-acetone solution is 0.6. Use the ternary phase
Equilibrium phase diagram of acetone/ethanol mixture under the pressure... | Download Scientific Diagram

Phase diagrams of acetonitrile or (acetone + water + EPPS) buffer phase separation systems at 298.15 K and quantum chemical modeling - ScienceDirect

SOLVED: The phase diagram for acetone (A) water (W) - methyl isobutyl ketone (MIK) 2 @ ternary mixture at 25 °C is given in the figure on the right. Note that the
![PDF] Liquid-Liquid Equilibrium Data for Butan-2-ol - Ethanol - Water, Pentan-1-ol - Ethanol - Water and Toluene - Acetone - Water Systems | Semantic Scholar PDF] Liquid-Liquid Equilibrium Data for Butan-2-ol - Ethanol - Water, Pentan-1-ol - Ethanol - Water and Toluene - Acetone - Water Systems | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/c1fa0b8a36ef8526c29d6a283bd62f1c9d37340c/2-Figure1-1.png)
PDF] Liquid-Liquid Equilibrium Data for Butan-2-ol - Ethanol - Water, Pentan-1-ol - Ethanol - Water and Toluene - Acetone - Water Systems | Semantic Scholar

Modeling of Mixing Acetone and Water: How Can Their Full Miscibility Be Reproduced in Computer Simulations? | The Journal of Physical Chemistry B

Water-ethanol-acetone phase diagram indicating the points corresponding... | Download Scientific Diagram
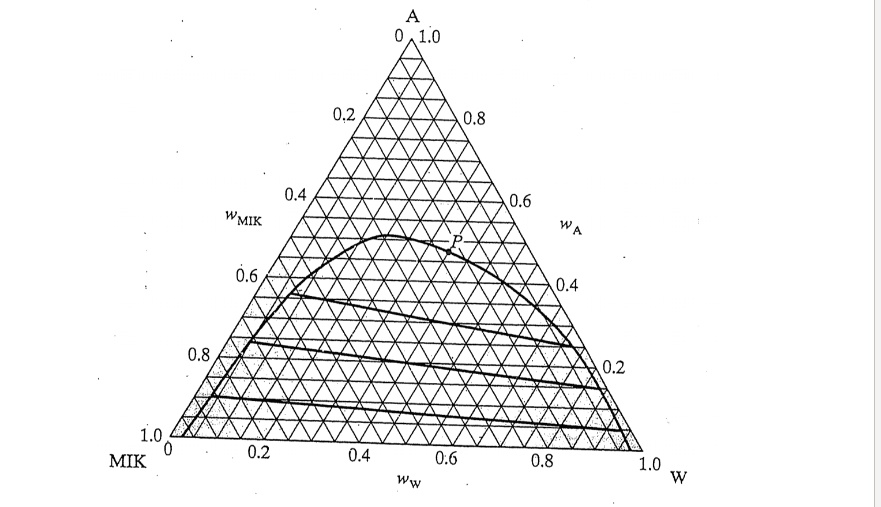
SOLVED: We wish to extract a mixture of 55 wt% acetone and 45 wt% water with methyl isobutyl ketone (MIK) at 298 K and 1 bar. If we contact 2 kg of
Water-ethanol-acetone phase diagram indicating the points corresponding... | Download Scientific Diagram


![Lecture 1 [PHY313] Lecture 1 [PHY313]](https://mini.physics.sunysb.edu/~marivi/TEACHING-OLD/PHY313/lib/exe/fetch.php?media=lectures:liquidphasediagram.png)